Timeline of experiment design
This is a timeline of experiment design, attempting to describe significant and illustrative events in the history of the field.
Sample questions
The following are some interesting questions that can be answered by reading this timeline:
Big picture
| Time period | Development summary | More details |
|---|---|---|
| Ancient Times – 1700s | Early foundations | Experimental thinking has ancient roots, notably in medicine and agriculture. The Earliest known clinical trial dates to 500 BCE, when Persian king Cyrus tests diets on his soldiers.[1] Greek physicians like Hippocrates[2] and Galen emphasize observation and rudimentary comparative methods. However, formal experimentation remains rare. The Scientific Revolution in the 1600s shifts focus from authority to empirical investigation. Francis Bacon promotes inductive reasoning and systematic observation, laying philosophical groundwork for modern science.[3] Nonetheless, experiments lack standardized design or statistical rigor. Scientists like Galileo[4] and Boyle[5] use controlled observations, but their methods are more qualitative than quantitative. The period ends with the emergence of probability theory by Pascal[6], Fermat[7], and Bernoulli[8], which begins to offer tools later essential for experimental statistics. |
| 1700s – Late 1800s | Classical period | The Age of Enlightenment brings advances in both scientific methodology and mathematical statistics.[9] Controlled experiments become more common in chemistry, physics, and agriculture. Notably, James Lind conducts one of the first controlled clinical trials in 1747, testing treatments for scurvy.[10] In parallel, Carl Linnaeus systematizes biological classification, influencing observational rigor.[11] Pierre-Simon Laplace and Carl Friedrich Gauss develop early statistical tools such as the normal distribution and least squares estimation, crucial for data analysis. Agricultural trials, especially in Europe, begin to use basic comparison and replication. However, randomness, replication, and formal control are not yet standardized. This period sets the stage for modern experimentation by linking empirical testing with early statistical theory, but experiments still lack formal design structures and reproducibility standards. The idea of a placebo effect—a therapeutic outcome derived from an inert treatment— is already discussed in 18th century psychology[12] |
| Early 1900s – 1950s | Formalization and statistics | This period marks the birth of modern experimental design. Ronald A. Fisher revolutionizes the field in the 1920s and 1930s with work at Rothamsted Experimental Station in England.[13] Fisher introduces core principles: randomization, replication, and blocking. He develops the analysis of variance (ANOVA) and emphasizes the role of statistical inference in drawing conclusions. His 1935 book, The Design of Experiments, would remain foundational. Experimental design becomes central to agriculture[14][14][15], biology, and later psychology and social sciences. Jerzy Neyman and Egon Pearson introduce hypothesis testing frameworks, further refining experimental methodology.[16] This era institutionalizes statistics as an essential scientific tool and formalizes the structure of experiments, enabling reproducibility and greater objectivity in scientific inference. The need to blind researchers becomes widely recognized in the mid-century.[17] |
| 1960s – present | Modern and computational era | Post-1950s, experimental design expand into diverse fields: psychology, economics, medicine, and engineering. With the rise of computers, simulation, factorial designs, and response surface methodology become widespread. Clinical trials adopt strict protocols, often randomized and double-blind, guided by ethical oversight. In social sciences, randomized controlled trials (RCTs) gain ground, supported by behavioral economics and development studies. The Bayesian approach gains traction, offering alternative inference frameworks. From the 1990s, machine learning and A/B testing transform experimental design in tech industries, allowing real-time, large-scale experimentation. Today, experimental design is deeply intertwined with data science, AI, and evidence-based policy, continually evolving to address high-dimensional data, ethical concerns, and reproducibility challenges across disciplines. |
Full timeline
| Year | Event type | Details | Geographical location |
|---|---|---|---|
| ca. 500 BCE | Experiment | Around this time, what would be often described as the earliest recorded experiment resembling a clinical trial occurs when King Nebuchadnezzar compares two diets—meat and wine versus beans and water—and observes that the group consuming the vegetarian diet appears healthier.[18] | Babylon (Mesopotamia) |
| ca. 400 BCE | Concept | Hippocrates promotes systematic observation and naturalistic explanations in medicine, emphasizing empirical study over supernatural interpretations.[18] | Greece |
| 1025 AD | Concept | Persian polymath Avicenna completes The Canon of Medicine, a major medical encyclopedia synthesizing Greco-Roman and Islamic knowledge. The work describes diseases, treatments, and hundreds of drugs, and outlines principles for experimentally testing medicines, including reproducibility of results, shaping medical education in both the Islamic world and Europe for centuries.[19] | Persia |
| 1620 | Concept | Francis Bacon publishes Novum Organum, proposing a new empirical approach to scientific inquiry. He argues that knowledge should be based on systematic observation and inductive reasoning rather than tradition or pure logic, helping establish methodological foundations for the modern scientific method and experimental science.[20] | England |
| 1683 | Method | Robert Boyle solidifies his reputation as a pioneer of modern experimental science by publishing New Experiments and Observations Touching Cold. During this period, Boyle and his contemporaries, including Robert Hooke, emphasize that chemical understanding must be grounded in controlled experimentation, precise documentation of apparatus, and the replication of results.[21] | England |
| 1700 | Concept | Korean mathematician Choi Seok-jeong is the first to publish an example of Latin squares of order nine, in order to construct a magic square, predating Leonhard Euler by 67 years.[22] Latin squares are used in combinatorics and in experimental design.[23] | Korea |
| 1713 | Concept | Jacob Bernoulli formulates the law of large numbers, establishing convergence of sample averages to expected values. | Switzerland |
| 1747 | Experiment | Scottish doctor James Lind conducts the first clinical trial when investigating the efficacy of citrus fruit in cases of scurvy. He randomly divides twelve scurvy patients, whose "cases were as similar as I could have them", into six pairs. Each pair is given a different remedy. According to Lind’s 1753 Treatise on the Scurvy in Three Parts Containing an Inquiry into the Nature, Causes, and Cure of the Disease, Together with a Critical and Chronological View of what has been Published of the Subject, the remedies were: one quart of cider per day, twenty-five drops of elixir vitriol (sulfuric acid) three times a day, two spoonfuls of vinegar three times a day, a course of sea-water (half a pint every day), two oranges and one lemon each day, and electuary, (a mixture containing garlic, mustard, balsam of Peru, and myrrh).[24] Lind would note that the pair who had been given the oranges and lemons were so restored to health within six days of treatment that one of them returned to duty, and the other was well enough to attend the rest of the sick.[24] | United Kingdom |
| 1747 | Experiment | James Lind conducts one of the earliest controlled clinical experiments on scurvy, dividing sailors into treatment groups to test dietary remedies, though without randomization.[25] | United Kingdom |
| 1784 | Experiment | The first blinded experiment is conducted by the French Academy of Sciences to investigate the claims of mesmerism as proposed by Franz Mesmer. In the experiment, researchers blindfolded mesmerists and asked them to identify objects that the experimenters had previously filled with "vital fluid". The subjects are unable to do so.[26] | France |
| 1798 | Statistical method | German mathematician Carl Friedrich Gauss develops the mathematical foundations of the method of least squares. Years later, the method would enable astronomers to predict the orbit of the asteroid Ceres after its discovery by Giuseppe Piazzi and help Franz Xaver von Zach successfully relocate it.[27] | Germany |
| 1815 | Concept | An article on optimal designs for polynomial regression is published by Joseph Diaz Gergonne.[28] | France |
| 1817 | Experiment | The first blinded experiment recorded outside of a scientific setting compares the musical quality of a Stradivarius violin to one with a guitar-like design. A violinist plays each instrument while a committee of scientists and musicians listen from another room so as to avoid prejudice.[29][30] | France |
| 1827 | Method | Pierre-Simon Laplace uses least squares methods to address analysis of variance problems regarding measurements of atmospheric tides.[31] | France |
| 1835 | Experiment | An early example of a double-blind protocol is the Nuremberg salt test performed by Friedrich Wilhelm von Hoven, Nuremberg's highest-ranking public health official.[32] | Germany |
| 1835 | Concept | Belgian statistician Adolphe Quetelet introduces the concept of the “average man” (l’homme moyen), arguing that human physical and social traits follow statistical distributions. By applying probability and quantitative analysis to crime, mortality, and demographics, he helps establish statistical approaches in sociology, demography, and the emerging social sciences.[33] | Belgium |
| 1860 | Method | German psychologist and physicist Gustav Theodor Fechner makes a groundbreaking contribution to the development of experimental psychology with the publication of Elements of Psychophysics. In this work, he seeks to test and justify the relationship between physical stimuli and the sensations they produce. Fechner proposes that mental experiences can be quantified by linking them to measurable physical changes, laying the foundation for psychophysics. Using experimental data, he formulates mathematical laws—most notably the Weber-Fechner law—that describe how perceived intensity varies with stimulus magnitude. His efforts help establish psychology as a quantitative science rooted in empirical observation.[34] | Germany |
| 1861 | Experiment | French chemist and microbiologist Louis Pasteur conducts controlled experiments demonstrating that microorganisms originate from airborne particles rather than spontaneous generation. In his memoir examining this doctrine, he shows that sterilized broth remain free of life unless exposed to contaminated air, helping establish the foundations of modern microbiology and germ theory.[35][36] | France |
| 1876 | Concept | American scientist Charles S. Peirce contributes the first English-language publication on an optimal design for regression models.[37] | United States |
| 1877 | Charles Sanders Peirce formalizes inquiry as a structured experimental process: hypotheses (abduction) generate testable predictions (deduction), which are evaluated through observation and experiment (induction). This integrates reasoning with empirical testing, establishing a cyclical, self-correcting model of experiment design focused on hypothesis testing and iterative refinement.[38] | ||
| 1879 | Organization | Experimental psychology emerges as a modern scientific discipline in Germany with the establishment of the first experimental laboratory by Wilhelm Wundt at the University of Leipzig. This marks a pivotal moment in the history of psychology, as Wundt seeks to separate psychology from philosophy by applying rigorous scientific methods. He introduces a mathematical and experimental approach to studying the human mind, emphasizing observation, measurement, and controlled experimentation. Wundt's work lays the foundation for psychology as an empirical science, influencing future researchers and schools of thought.[34] | Germany |
| 1882 | Concept | In his published lecture at Johns Hopkins University, Peirce introduces experimental design with these words:
|
United States |
| 1885 | Concept | Analysis of variance. An eloquent non-mathematical explanation of the additive effects model becomes available.[40] | United Kingdom |
| 1880s | Experiment | Charles Sanders Peirce and Joseph Jastrow introduce randomized experiments in the field of psychology.[41] | United States |
| 1897 | Experiment | Norman Triplett conducts one of the first social psychology experiments on cyclist performance.[42] | United States |
| 1900 | Method | The P-value is first formally introduced by Karl Pearson, in his Pearson's chi-squared test, using the chi-squared distribution and notated as capital P.[43] Since then, P-values would become the preferred method to summarize the results of medical articles.[44][45] | United Kingdom |
| 1903 | Concept | American physician Richard Clarke Cabot concludes that the placebo should be avoided because it is deceptive.[46] | United States |
| 1907 | Experiment | The first study recorded to have a blinded researcher is conducted by W. H. R. Rivers and H. N. Webber to investigate the effects of caffeine.[47] | United Kingdom |
| 1908 | Method | British statistician William Sealy Gosset, working at Guinness, introduces the Student’s t-distribution to analyze small sample data when population variance is unknown. Publishing under the pseudonym “Student” in the journal Biometrika, he provides a statistical method widely used for inference with limited data.[48] | United Kingdom |
| 1910s | Concept | Ronald A. Fisher develops the concept of variance as a measure of population variability, laying the foundation for later statistical methods such as analysis of variance (ANOVA).[49] | United Kingdom |
| 1918 | Concept | English statistician Ronald Fisher introduces the term variance and proposes its formal analysis in his article The Correlation Between Relatives on the Supposition of Mendelian Inheritance.[50] | United Kingdom |
| 1918 | Method | Kirstine Smith publishes a major study in Biometrika analyzing the statistical precision of polynomial regression estimates. She derives principles for choosing observation points that minimize estimation variance, introducing foundational methods for optimal experimental design in polynomial models and influencing later developments in statistical design theory.[51] | United Kingdom |
| 1918 | Method | Optimal designs for polynomial regression formally developed | Denmark (Kirstine Smith) |
| 1918–1940s | Method | Ronald A. Fisher and collaborators establish the foundations of modern experimental design in agricultural research, introducing factorial designs and analysis of variance (ANOVA).[14] | United Kingdom |
| 1919 | Organization | Ronald A. Fisher is hired as a statistician at the Rothamsted Experimental Station, where his work on poorly designed agricultural data helps trigger the modern statistical approach to experimental design.[49] | United Kingdom |
| 1919 | Method | R. A. Fisher at the Rothamsted Experimental Station in England starts developing modern concepts of experimental design in the planning of agricultural field experiments.[52] | England |
| 1921 | Method | Ronald Fisher publishes his first application of the analysis of variance.[53] | United Kingdom |
| 1920s | Method | Ronald Fisher develops randomized block design and formal principles of blocking to control variation in experiments. | United Kingdom |
| 1920s | Method | Fisher introduces key principles of modern experimental design, including confounding, randomization, replication, blocking, Latin square designs, and other factorial designs.[49] | United Kingdom |
| 1920s | Method | analysis of variance and maximum likelihood estimation are developed as major statistical tools for analyzing experimental data.[49] | United Kingdom |
| 1920s | Organization | The first academic statistics department is established at Iowa State University under George Snedecor, helping institutionalize statistical experimental methods.ref name="MurphyHistoryED"/> | United States |
| 1920s | Method | George Snedecor develops the F-test and F-distribution, which become central tools for decisions in ANOVA-based experimental designs.[49] | United States |
| 1920s–1930s | Method | Ronald A. Fisher establishes the foundations of modern design of experiments, introducing randomization, replication, blocking, factorial designs, likelihood methods, and analysis of variance (ANOVA).[54] | United Kingdom |
| 1920s–1930s | Concept | Fisher formalizes the principle that well-designed experiments are essential for valid statistical inference, shifting focus from analysis to experimental planning.[55] | United Kingdom |
| 1920s–early 1930s | Method | Ronald A. Fisher establishes the foundations of statistical design, introducing randomization, replication, blocking, factorial design, and analysis of variance.[55] | United Kingdom |
| 1923 | Field development | The first randomization model is published in Polish by Jerzy Neyman.[56] | Poland |
| 1925 | Method | Ronald Fisher publishes Statistical Methods for Research Workers, developing tests of statistical significance, proposing the 0.05 threshold for hypothesis testing, and popularizing analysis of variance as a method for partitioning variation in experimental data.[57][58][59][60][61] | United Kingdom |
| 1925 | Publication | Fisher publishes Statistical Methods for Research Workers, promoting statistical methods for analyzing experimental data.[54] | United Kingdom |
| 1925 | Literature | British statistician Ronald Fisher publishes Statistical Methods for Research Workers, which is considered a seminal book in which he explains the concept of statistical significance.[62] | United Kingdom |
| 1925 | Publication | Fisher publishes Statistical Methods for Research Workers, one of the foundational texts of modern statistical analysis of experiments.[49] | United Kingdom |
| 1925–1926 | Concept | Ronald Fisher states core principles of modern experimental design, including meaningful estimation of experimental variability, randomization, blocking, and factorial experimentation.[63] | United Kingdom |
| 1926 | Method | Ronald Fisher advocates the use of factorial experiments in The Arrangement of Field Experiments, showing that studying multiple factors simultaneously is more efficient than varying one factor at a time.[64] | United Kingdom |
| 1926 | Publication | John Russell (agricultural scientist) publishes "Field Experiments: How They Are Made and What They Are", summarizing contemporary practices and principles of experimental design in agricultural research.[65] | United Kingdom |
| 1930s | Method | Ronald Fisher and others formalize fractional factorial design to study multiple factors efficiently with fewer runs. | United Kingdom |
| 1930s | Concept | The size of an experiment is recognized as a key design decision, with sample size linked to precision, significance, and statistical power.[63] | Global |
| 1930s | Concept | The advantages of factorial experimentation over one-factor-at-a-time methods are articulated, especially its economy of resources and ability to estimate interrelationships among factors.[63] | Global |
| 1930s | Field development | Early applications of statistical experimental design begin to appear in industrial settings, extending beyond agriculture.[55] | Global |
| 1930s | Method | Randomization is formalized as the use of random numbers or equivalent devices to assign treatments and reduce bias from extraneous variation.[63] | Global |
| 1930s | Concept | Experimental error is formally distinguished as a major obstacle to precision, arising from extraneous sources of variation beyond the treatments themselves.[63] | Global |
| 1930s | Concept | Bias is distinguished from random error, emphasizing that systematic error can mislead conclusions and often cannot be detected through statistical analysis alone.[63] | Global |
| 1930s | Method | double-blind experiments and placebo controls are recognized as important safeguards against bias in experiments involving subjective or clinical judgments.[63] | Global |
| 1930s | Method | Refinements of experimental technique, such as practice runs, clearer instructions, environmental control, and improved measurement instruments, are emphasized as ways to reduce experimental error.[63] | Global |
| 1930s | Method | blocking is presented as a method for increasing experimental precision by balancing treatments across subjects or units with similar initial characteristics.[63] | Global |
| 1930s | Method | matched pairs design and matched groups designs are used with human subjects as forms of blocking analogous to blocks in agricultural experiments.[63] | Global |
| 1930s | Concept | Core principles of experimental design—randomization, replication, and blocking—are established as fundamental techniques to reduce bias and improve precision in experiments.[25] | United Kingdom |
| 1930s | Method | analysis of covariance is introduced as a more accurate statistical adjustment for initial differences when blocking is not feasible or when further precision is needed.[63] | Global |
| 1930s | Method | factorial experimentation is formalized as a way to investigate several factors simultaneously, improving efficiency and enabling study of interactions among variables.[63] | Global |
| 1934 | Publication | Fisher publishes The Design of Experiments, formalizing principles that become the basis of modern prospective experimental research.[49] | United Kingdom |
| 1935 | Publication | Ronald Fisher publishes The Design of Experiments, a foundational work that formalizes principles such as randomization, replication, and blocking, and emphasizes the importance of efficient experimental design.[66][67][68][69] | United Kingdom |
| 1935 | Method | Ronald A. Fisher publishes The Design of Experiments, formalizing the modern statistical framework of experimental design and introducing key principles such as randomization, replication, and blocking.[25] | United Kingdom |
| 1935 | Method | Fisher shows that randomization can justify valid tests of treatment effects without strong assumptions about the underlying frequency distribution of the data.[63] | United Kingdom |
| 1935 | Publication | Fisher publishes The Design of Experiments, formalizing principles that become the basis of modern experimental science. [54] | United Kingdom |
| 1935 | Publication | Fisher presents these principles more fully in The Design of Experiments, consolidating the modern framework for statistical experimental design.[63] | United Kingdom |
| 1937 | Experiment | First large-scale randomized experiment in education conducted by the U.S. Office of Education. | United States |
| 1939 | Concept | A publication by Bose and Nair underlie the concept of association scheme. In their paper, they introduced the concept of association schemes as a way to study the structure of contingency tables. They show that association schemes can be used to represent the dependencies between the variables in a contingency table, and that they can be used to derive statistical tests for independence.[70] | India |
| 1940 | Method | Raj Chandra Bose and K. Kishen at the Indian Statistical Institute independently find some efficient designs for estimating several main effects. | India |
| 1940s | Method | George Box and collaborators develop early central composite design methods for response surface optimization. | United Kingdom |
| 1940s | Method | sequential analysis emerges during World War II to improve efficiency in military experimentation and decision-making.[14] | United States |
| 1940s | Method | Development of orthogonal designs and Latin squares as structured experimental layouts for controlling variation in experiments.[14] | United Kingdom |
| 1946 | Method | R.L. Plackett and J.P. Burman publish a renowned paper titled "The Design of Optimal Multifactorial Experiments". The paper introduces what would be called Plackett–Burman designs, which are highly efficient screening designs with run numbers that are multiples of four. These designs are particularly useful for experiments where only main effects are of interest. In a Plackett-Burman design, main effects are often heavily confounded with two-factor interactions, making them suitable for screening experiments. For instance, a Plackett-Burman design with 12 runs can be utilized for an experiment containing up to 11 factors.[71] | United Kingdom |
| 1948 | Experiment | The first modern randomized controlled trial is conducted in the streptomycin study by the Medical Research Council (United Kingdom). | United Kingdom |
| 1948 | Experiment | The Medical Research Council conducts a landmark randomized controlled trial of streptomycin for pulmonary tuberculosis, establishing modern clinical trial methods such as random allocation and blinding. Results show reduced mortality but emerging drug resistance and side effects, highlighting need for combination therapies and shaping evidence-based medicine future practice.[72] | United Kingdom |
| 1948 | Concept | British statistician Frank Yates introduces the concept of restricted randomization.[73][74] | United Kingdom |
| 1948 | Experiment | The randomized streptomycin trial for tuberculosis establishes one of the first modern randomized controlled clinical trials | United Kingdom |
| 1949 | Method | Genichi Taguchi begins developing his experimental design techniques while working at Japan’s Electrical Communications Laboratories (ECL) in the post–World War II reconstruction period. Tasked with improving research and development productivity, he formulates a new approach to quality improvement that emphasizes off-line quality control, robust design, and statistical experimentation. These early efforts lay the foundations of what would later become known as the Taguchi Methods, integrating engineering design with statistical principles to systematically reduce variability, improve product quality, and lower societal and manufacturing costs.[75] | Japan |
| Mid-20th century | Method | Standardized procedural steps for experimental design are established, including problem definition, variable selection, factor identification, experimental execution, and statistical analysis.[25] | Global} |
| Mid-20th century | Method | analysis of variance (ANOVA) becomes the central statistical framework for analyzing experimental data and guiding experimental design decisions.[25] | Global |
| 1950 | Publication | Gertrude Mary Cox and William Gemmell Cochran publish the book Experimental Designs, which would become the major reference work on the design of experiments for statisticians for years afterwards.[76] | United States |
| 1950 | Publication | Cochran and Cox provide more accurate tables and methods for sample size determination in experimental design.[63] | United States |
| 1950 | Method | Cochran and Cox formalize principles of sample size determination in experimental design. | United States |
| 1950 | Concept | Randomized controlled trials begin to emerge as the gold standard in medical research, enabling systematic causal inference | Global |
| 1950s | Method | Statistical methods and experimental design spread rapidly through the medical literature, extending techniques developed for agriculture into biomedical research.[49] | Global |
| 1950s | Method | Factorial experimental designs become widely used in industrial experimentation to study multiple variables simultaneously | United States |
| 1950s | Method | Control groups are emphasized as essential in evaluatory studies for distinguishing treatment effects from other changes over time.[63] | Global |
| 1950s–1970s | Field development | Statistical experimental design expands from agriculture into industrial applications, particularly in chemical and process industries.[54] | Global |
| 1951 | Method | Formal placebo-controlled and multi-arm clinical trial designs are developed to improve reliability of treatment comparisons | United States |
| 1951 | Method | George E. P. Box and K. B. Wilson introduce response surface methodology (RSM), using sequences of designed experiments and second-degree polynomial models to approximate and optimize responses, even with limited process knowledge.[77][14] | United Kingdom |
| 1951 | Method | George E. P. Box and K. B. Wilson introduce response surface methodology (RSM), enabling optimization of industrial processes through sequential experimentation.[54] | United Kingdom |
| 1951 | Method | George E. P. Box and K. B. Wilson introduce response surface methodology, enabling sequential and adaptive optimization in industrial experiments.[55] | United Kingdom |
| 1952 | Concept | American mathematician and statistician Herbert Robbins recognizes the significance of a problem where a gambler faces a trade-off between "exploitation" of the machine with the highest expected payoff and "exploration" to learn about other machines' payoffs. This problem involves pulling levers on different machines, each providing random rewards from unknown probability distributions. The gambler aims to maximize the total rewards earned over a sequence of lever pulls. Robbins devised convergent population selection strategies in his work on "some aspects of the sequential design of experiments."[78] | United States |
| 1952 | Concept development | Bose and Shimamoto introduce the term association scheme.[79] | United States |
| 1954 | Experiment | The Salk polio vaccine trial becomes one of the first large-scale randomized controlled trials, involving over one million participants | United States |
| 1954 | Concept | Paul Lazarsfeld and others formalize factor analysis as a tool for latent variable modeling in experiments. | United States |
| 1954 | Publication | Edwin Boring discusses the history and meanings of control treatments, clarifying the role of controls in experimental research.[63] | United States |
| 1954 | Publication | American experimental psychologist Edward Boring writes an article titled The History of Experimental Design. In this article, Boring notes that the early history of ideas on the planning of experiments has been "but little studied".[52] | United States |
| 1955 | Experiment | An influential study entitled The Powerful Placebo firmly establishes the idea that placebo effects are clinically important.[80] | United States |
| 1950s | Method | W. Edwards Deming promotes statistical quality control in Japan, initiating the quality revolution and widespread industrial use of experimental design.[14] | Japan |
| 1950s–1980s | Field development | Response surface methods and statistical design techniques spread across chemical and process industries, especially in research and development.[55] | Global |
| 1959–1961 | Method | Jack Kiefer and Jacob Wolfowitz develop formal optimal design theory, introducing criteria-based selection of experimental designs for maximum precision.[55] | United States |
| 1960s | Concept | Increasing emphasis on internal validity leads to stricter control of confounding variables in experimental design | Global |
| 1960s | Method | Double-blind experimental designs become standard in clinical trials to reduce bias from participants and researchers | Global |
| 1960s | Method | Japanese industries adopt experimental design and statistical quality control, leading to major improvements in manufacturing quality and global competitiveness.[14] | Japan |
| 1960s | Experiment | randomized controlled trial becomes the gold standard in clinical research, replacing anecdotal medical evidence with controlled experimental methods.[14] | United States |
| 1960 | Industrial statistics | Japanese engineer and statistician Genichi Taguchi publishes Design of Experiments for Engineers, advancing statistical methods for industrial experimentation. His work helps formalize what later would become known as Taguchi methods, integrating experimental design and statistical analysis to improve product quality, optimize manufacturing processes, and reduce costs in engineering and industry.[81] | Japan |
| 1960 | Method | George E. P. Box and Donald Behnken devise what is statistics is known as Box–Behnken designs, which are experimental designs for response surface methodology[82] | United States |
| 1961 | Concept | Leslie Kish introduces the term design effect.[83] | United States |
| 1961 | Concept | The term nocebo (Latin nocēbō, "I shall harm", from noceō, "I harm")[84] is coined by Walter Kennedy to denote the counterpart to the use of placebo (Latin placēbō, "I shall please", from placeō, "I please"; a substance that may produce a beneficial, healthful, pleasant, or desirable effect). Kennedy emphasized that his use of the term "nocebo" refers strictly to a subject-centered response, a quality inherent in the patient rather than in the remedy".[85] | United States |
| 1962 | Policy | The Kefauver Harris Amendment requires proof of efficacy through controlled trials before drug approval, institutionalizing experimental design in regulation. | United States |
| 1962 | Experiment | Vernon L. Smith publishes An Experimental Study of Competitive Market Behavior in the Journal of Political Economy. Using controlled laboratory market experiments with human participants, he tests hypotheses of neoclassical competitive theory and demonstrates price convergence toward equilibrium, helping establish experimental economics as a systematic empirical research methodology.[86] | United States |
| 1962 | Method | British statistician John Nelder proposes a set of systematic, circular experimental designs as an alternative to the replicated, full factorial spacing experiments. These designs, known as the Nelder 'wheel' design, are developed to address limitations related to space and plant material. The design consists of a circular plot with concentric circumferences radiating outward, connected by spokes that extend from the center to the farthest circumference. Trees are planted at the intersections of spokes and circumferences within the plot.[87] | United Kingdom |
| 1963 | Concept | Campbell and Stanley discuss design according to the categories of preexperimental designs, experimental designs, and quasi-experimental designs.[88] | United States |
| 1960s | Concept | The concept of instrumental variable methods (instrument effects) is formalized in econometrics. | United States |
| 1960s | Method | quasi-experimental design is formalized by Donald T. Campbell for causal inference without randomization. | United States |
| 1960s–1970s | Concept | Industrial experiments are recognized as distinct due to immediacy and sequential learning, enabling rapid iteration and adaptive experimentation.[55] | Global |
| 1965 | Method | Leslie Kish develops modern survey sampling theory and introduces design-based inference. | United States |
| 1970 | Concept | Early formulation of the potential outcomes framework introduces counterfactual reasoning in causal analysis | United States |
| 1970 | Method | system identification methods emerge in engineering for modeling dynamic systems from data. | United States |
| 1970s | Method | computer experiment methodology emerges for studying simulation models. | United States |
| 1970s | Method | Multi center clinical trials become standard, enabling large-scale randomized evaluation across diverse populations | Global [89] |
| 1970s | Method | Response surface methodology is widely applied in industrial process optimization and product design | United States [90] |
| 1970s–1980s | Method | Genichi Taguchi develops robust parameter design methods to reduce variability and improve product quality.[14] | Japan |
| 1970s–1990 | Method | Quality improvement methodologies such as Total Quality Management (TQM) and Continuous Quality Improvement (CQI) integrate experimental design into industrial processes.[14] | Global |
| 1972 | Publication | Herman Chernoff writes an overview of optimal sequential designs[91] In the design of experiments, optimal designs is a class of experimental designs that are optimal with respect to some statistical criterion. | United States |
| 1974 | Concept | :contentReference[oaicite:0]{index=0} formalizes the potential outcomes framework, defining causal effects using counterfactuals | United States |
| 1976 | Publication | Douglas C. Montgomery publishes Design and Analysis of Experiments, a comprehensive textbook on the design and analysis of experiments. The book covers a wide range of topics, including principles of experimental design, different types of experimental designs, analysis of experimental data, and use of experimental design in a variety of fields, such as agriculture, industry, and medicine.[92] | United States |
| 1977 | Method | The concept of Pocock boundary is introduced by the medical statistician Stuart Pocock.[93] | United Kingdom |
| 1977 | Method | :contentReference[oaicite:1]{index=1} develops response surface methodology to optimize industrial experiments | United Kingdom |
| 1977 | Method | Stuart Pocock introduces group sequential designs for clinical trials. | United Kingdom |
| 1978 | Concept | According to Box et al., experimental design refers to the systematic layout of combinations of variables. The layouts in the case of concepts are test concepts or test vignettes.[94] | United States |
| 1978 | Concept | Ulrich Krengel and Louis Sucheston (with David J. H. Garling) formulated the Prophet Inequality in optimal stopping theory. They show that a gambler observing sequential random rewards can secure at least half the expected payoff of a “prophet” who knows all outcomes in advance, establishing a foundational result in probability theory and decision processes.[95] | United States |
| 1979 | Method | Marvin Zelen publishes his new method, which would later be called Zelen's design.[96][97] | United States |
| 1979 | Method | Michael McKay at Los Alamos National Laboratory makes a significant contribution to the field of statistical sampling by introducing the concept of latin hypercube sampling.[98] | United States |
| Late 1970s | Method | Genichi Taguchi promotes robust parameter design, emphasizing quality improvement and reduction of variability using orthogonal arrays and fractional factorial designs.[54] | Japan |
| Late 1970s | Method | Genichi Taguchi introduces robust parameter design, emphasizing reduction of variability and improvement of product quality under noise conditions.[55] | Japan |
| Late 1970s–1980s | Field development | Growing interest in quality improvement in Western industry leads to broader adoption of experimental design methods.[55] | Global |
| 1980 | Concept | Greater attention to external validity emphasizes the generalizability of experimental findings | Global |
| 1980 | Concept | Alan E. Kazdin classifies research designs into experimental, quasi-experimental, and correlational categories, clarifying distinctions in causal inference and methodological rigor. | United States |
| 1980s | Method | Genichi Taguchi introduces robust design methods to improve product quality by reducing sensitivity to variability in manufacturing processes. | Japan |
| 1980s | Method | supersaturated designs are developed for screening large numbers of factors. | United States |
| 1980s | Concept | manipulation check becomes standard in experimental psychology to validate treatment implementation. | United States |
| 1980s | Method | Quality engineering approaches emphasize robustness and variance reduction in manufacturing processes | Japan [99] |
| 1980s | Method | Meta analysis emerges as a method to systematically combine results from multiple experiments | Global |
| 1980s | Field development | Designed experiments become widely adopted in manufacturing industries (automotive, aerospace, electronics), driven by quality improvement initiatives.[54] | Global |
| 1980s | Method | Large-scale randomized trials such as the ISIS heart studies demonstrate the feasibility of very large clinical experiments | Global [100] |
| 1981 | Allen Neuringer first proposes the idea of using single case designs (sometimes referred to as n-of-1 trials) for self-experimentation.[101] | ||
| 1982 | Literature | British statistician George Box publishes Improving Almost Anything: Ideas and Essays, which gives many examples of the benefits of factorial experiments.[102] | |
| 1984 | Concept | Stuart Hurlbert publishes a paper in Ecological Monographs where he analyzes 176 experimental studies in ecology. He discovers that 27% of these studies suffer from 'pseudoreplication,' meaning they use statistical testing in situations where treatments are not replicated or replicates were not independent. When considering only studies that use inferential statistics, the percentage of pseudoreplication increases to 48%. To address this issue, Hurlbert suggests interspersing treatments in experiments, even if it means sacrificing randomized samples, particularly in smaller experiments. This approach aims to overcome the problem of pseudoreplication in ecological studies.[103] | United States |
| 1986 | Experiment | Robert LaLonde finds that findings of econometric procedures assessing the effect of an employment program on trainee earnings do not recover the experimental findings. This is considered to be the start of experimental benchmarking in social science. [104] | United States |
| 1986 | Concept | Fred N. Kerlinger describes the MAXMINCON principle, emphasizing maximizing systematic variance, controlling extraneous variance, and minimizing error variance in experimental design.[88] | United States |
| 1986 | Concept | Selection bias is formally recognized as a central limitation of observational and non-randomized studies | Global |
| 1987 | Publication | Australian mathematician Anne Penfold Street publishes Combinatorics of Experimental Design, a textbook on combinatorial methods in experimental design.[105] | Australia |
| 1988 | Publication | Roger Mead publishes The Design of Experiments: Statistical Principles for Practical Applications, a textbook presenting practical principles of experimental design and analysis.[106] | United Kingdom |
| 1989 | Publication | Perry D. Haaland publishes Experimental Design in Biotechnology, presenting statistical experimental design and analysis as a problem-solving tool in biotechnology.[107][108][109] | United States |
| 1989 | Publication | Jerome Sacks and collaborators discuss statistical issues in the design and analysis of computer and simulation experiments, helping establish the field of computer experiments.[110] | United States |
| Late 1980s | Concept | Critical evaluation of Taguchi methods leads to refinement of statistical foundations and development of alternative robust design methodologies.[54] | Global |
| 1990 | Concept | The term evidence-based medicine is introduced, promoting the systematic use of randomized evidence in clinical decision making | Canada [111] |
| 1990s | Method | Six Sigma integrates statistical experimental design into industrial quality control and process improvement,[14] | United States |
| 1990s | Method | optimal design approaches gain prominence, focusing on maximizing information and efficiency under resource constraints, supported by advances in computing.[54] | Global |
| 1990s | Field development | Designed experiments expand into new domains including services, finance, and government operations.[54] | Global |
| 1990s | Method | The modern era of experimental design begins, driven by globalization and economic competition, expanding DOE across industries and services.[14] | Global |
| 1990s–present | Field development | The Optimal Design Era emerges, characterized by widespread use of computational algorithms, software tools, and expansion of DOE across diverse sectors.[54] | Global |
| 1991 | Organization | The first International Data Farming Workshop takes place. Since then, 16 additional workshops would be held. These workshops would witness broad participation from various countries, including Canada, Singapore, Mexico, Turkey, and the United States.[112] | United States |
| 1994 | Method | The Neyer-d optimal test is first described by Barry T. Neyer.[113] | United States |
| 1995 | Method | Instrumental variable techniques become widely used to estimate causal effects from observational data | United States |
| 1995 | Method | Chris Nachtsheim and Ruth Meyer introduce the coordinate exchange algorithm, enabling computational generation of optimal experimental designs.[54] | United States |
| 1996 | Ethical oversight | International Conference on Harmonisation (ICH) guidelines for Good Clinical Practice (GCP) established.[114] | Global |
| 1996 | Policy | The CONSORT statement is introduced to improve reporting quality of randomized controlled trials. Published in major medical journals, it provides a standardized checklist and flow diagram to ensure transparency, completeness, and methodological clarity, addressing widespread deficiencies in trial reporting and establishing a foundation for evidence-based clinical research standards.[115] | Global |
| 1998 | Organization | Stat-Ease releases its first version of Design–Expert, a statistical software package specifically dedicated to performing design of experiments.[116] | United States |
| 1999 | Concept | Basili et al use the term family of experiments to refer to a group of experiments that pursue the same goal and whose results can be combined into joint—and potentially more mature—findings than those that can be achieved in isolated experiments.[117] | United States |
| 2000 | Concept | Judea Pearl develops causal graphical models, enabling formal reasoning about causality. | United States |
| 2000 (January 19) | Publication | A First Course in Design and Analysis of Experiments.[118] | United States |
| Late 1990s | Organization | Bradley Jones joins JMP (software), contributing to the development of advanced DOE software tools for engineers and researchers.[54] | United States |
| Late 20th century | Concept | Fisher’s principles of randomization, replication, and blocking become standard features of statistically rigorous experiments in the biological and biomedical sciences.[49] | Global |
| Late 20th century | Concept | Recognition that all experiments are inherently designed, with emphasis on the importance of proper planning to avoid wasted resources and invalid results.[55] | Global |
| Late 20th century | Method | Advances in computing and algorithms enable practical implementation of optimal design, expanding its use in scientific and industrial applications.[55] | Global |
| 2000s | Method | Large-scale online randomized experiments become standard in technology companies for product optimization | United States |
| 2000 | Concept | Judea Pearl formalizes causal graphical models, advancing causal inference theory. | United States |
| 2000s | Method | experimetrics emerges in economics, applying experimental methods to causal policy evaluation. | United States |
| 2000s | Method | A/B testing becomes standard in digital platforms for continuous experimentation. | United States |
| 2000s | Method | Online randomized controlled experiments (A/B testing) become standard in internet companies for product and interface optimization, enabling continuous data-driven decision making.[119] | United States |
| 2000s | Method | Systematic reviews and trial registries improve transparency and reduce publication bias in experimental research | Global |
| 2000s | Concept | Comparative effectiveness research expands use of experiments to evaluate real world treatment outcomes | United States |
| 2000s | Method | Bradley Jones and Chris Nachtsheim develop definitive screening designs, enabling efficient identification of important factors with minimal runs.[54] | United States |
| 2000s | Method | Bradley Jones introduces Bayesian D-optimal design approaches, incorporating prior knowledge into experimental planning.[54] | United States |
| 2000s | Method | Bradley Jones develops the Custom Design platform, enabling flexible, computer-generated optimal experimental designs tailored to specific problems.[54] | United States |
| 2000s | Method | Advances in supersaturated designs and split-plot designs improve efficiency in experiments with many factors and constraints.[54] | United States |
| 2000s–present | Method | Development of group-orthogonal supersaturated designs (GO-SSDs) improves factor screening when variables exceed experimental runs.[54] | United States |
| Late 20th century | Application | Experimental design expands beyond agriculture into engineering, business, and scientific research, becoming a general-purpose methodology.[25] | Global |
| 21st century | Application | Designed experiments are widely applied across service sectors including finance, business operations, and government, reflecting the broad adoption of statistical experimentation.[25] | Global |
| 2001 | Method | Daniel Kahneman initiates the practice of adversarial collaboration.[120] | United States |
| 2002 | Concept | The terms exploratory thought and confirmatory thought are introduced by social psychologist Jennifer Lerner and psychology professor Philip Tetlock in their book Emerging Perspectives in Judgment and Decision Making.[121] | United States |
| 2003 | Organization | Abdul Latif Jameel Poverty Action Lab is founded to scale randomized evaluations in development economics | United States |
| 2003 | Organization | The Abdul Latif Jameel Poverty Action Lab is founded to scale randomized evaluations in development economics. [122] | United States |
| 2005 | Experiment | Study determines that most clinical trials have unclear allocation concealment in their protocols, in their publications, or both.[123] | United Kingdom |
| 2007 | Organization | :contentReference[oaicite:0]{index=0} scales A B testing infrastructure to run thousands of experiments annually | United States |
| 2009 | Method | Adversarial collaboration is recommended by Daniel Kahneman[124] and others as a way of resolving contentious issues in fringe science, such as the existence or nonexistence of extrasensory perception.[125] | United States |
| 2010 | Concept | Asbjørn Hróbjartsson and Peter C. Gøtzsche argue in a meta-analysis that observed placebo effects can arise from bias due to lack of blinding, emphasizing the importance of proper control and blinding in experimental design.[126] | Denmark |
| 2010 | Organization | :contentReference[oaicite:4]{index=4} expands the global use of randomized evaluations in public policy | Global |
| 2010s | Method | Adaptive experimental designs allow modification of trials based on interim results while maintaining statistical validity | Global |
| 2010s | Method | Continuous A/B testing systems enable rapid, large-scale experimentation in digital platforms, allowing ongoing optimization based on user behavior data.[127] | Global |
| 2010s | Method | Large scale randomized evaluations are used to test education, health, and poverty interventions in developing countries | Global |
| 2010s | Method | Platform trials enable simultaneous testing of multiple interventions under a unified experimental framework | Global |
| 2014 | A study by Nosek and Lakens finds that preregistered studies are more likely to replicate than non-preregistered studies.[128] | ||
| 2015 | Method | Bayesian approaches to experimentation gain adoption in industry and online experimentation | Global |
| 2018 | Concept | Increased focus on heterogeneous treatment effects highlights variation in causal effects across subpopulations | Global |
| 2019 | Recognition | Nobel Prize in Economics awarded to Abhijit Banerjee, Esther Duflo, and Michael Kremer for experimental approaches to alleviating global poverty | Sweden [129] |
| 2019 | Policy | The U.S. Food and Drug Administration provides guidance on the use of adaptive designs in clinical trials, formalizing regulatory standards for flexible experimental designs.[130] | United States |
| 2020s | Method | Integrated experimentation platforms enable continuous large-scale randomized testing in digital environments | Global |
| 2020s | Concept | Causal machine learning combines statistical learning with causal inference to estimate complex treatment effects | Global |
| 2020s | Method | Machine learning is integrated with experimentation to estimate heterogeneous treatment effects and optimize interventions | Global [131] |
Numerical and visual data
Google Scholar
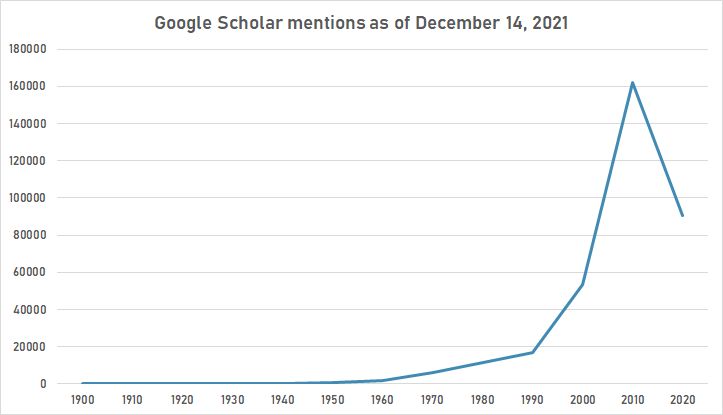
The following table summarizes per-year mentions on Google Scholar as of December 14, 2021.
| Year | "experimental design" |
|---|---|
| 1900 | 30 |
| 1910 | 17 |
| 1920 | 13 |
| 1930 | 19 |
| 1940 | 62 |
| 1950 | 425 |
| 1960 | 1,590 |
| 1970 | 6,240 |
| 1980 | 11,400 |
| 1990 | 17,000 |
| 2000 | 53,200 |
| 2010 | 162,000 |
| 2020 | 90,600 |
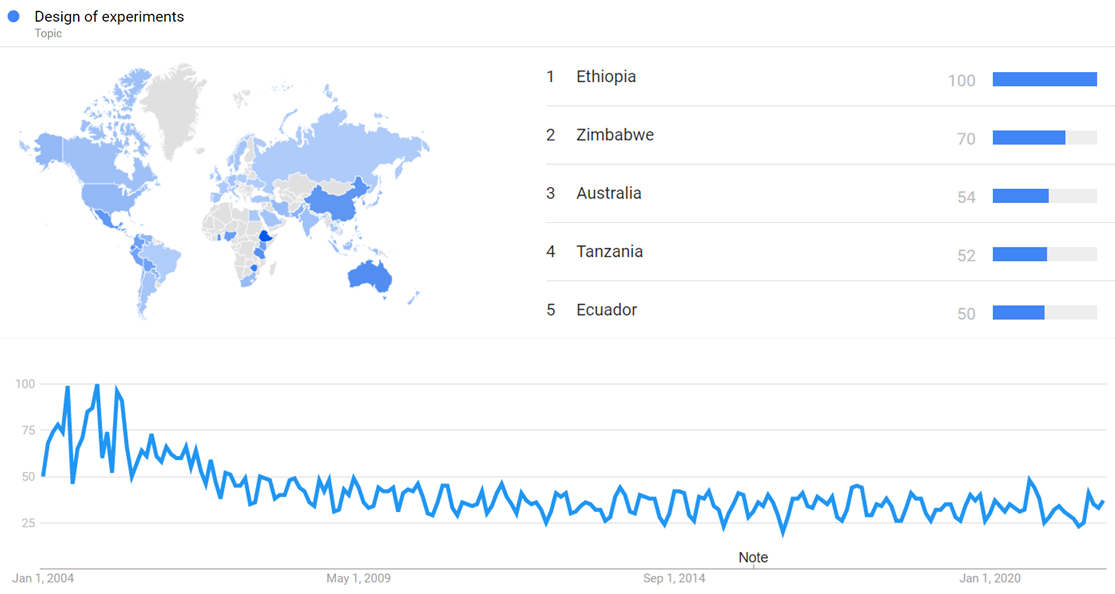
Google Trends
The chart below shows Google Trends data for Design of experiments (Topic), from January 2004 to December 2021, when the screenshot was taken. Interest is also ranked by country and displayed on world map.[132]
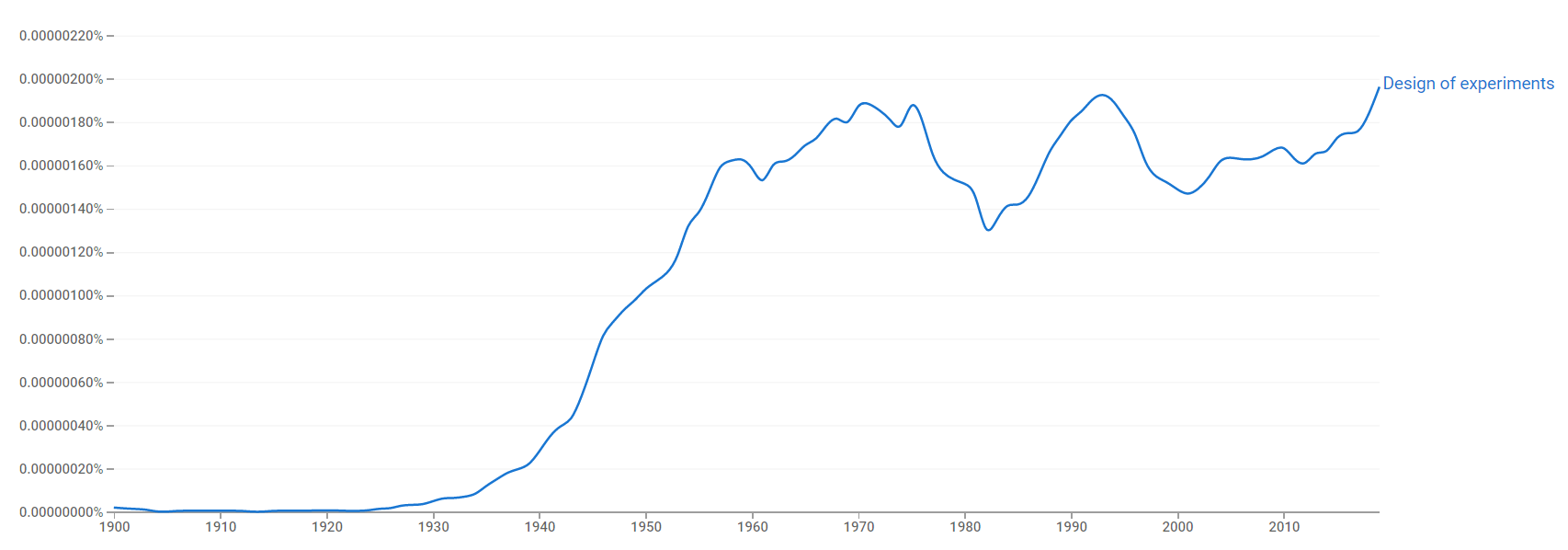
Google Ngram Viewer
The chart below shows Google Ngram Viewer data for Design of experiments, from 1900 to 2019.[133]
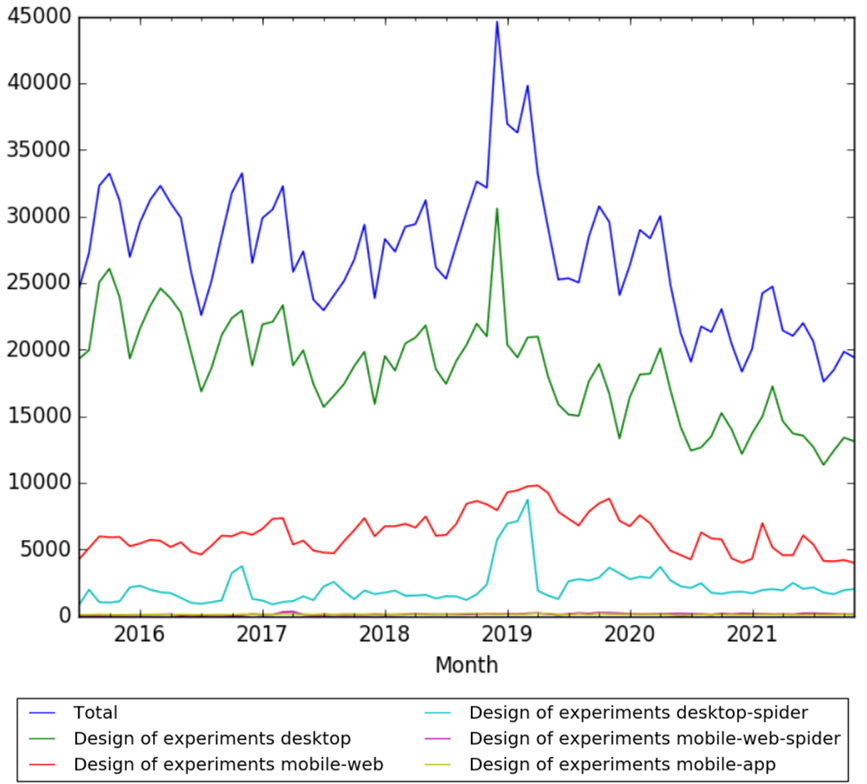
Wikipedia Views
The chart below shows pageviews of the English Wikipedia article Design of experiments, from July 2015 to November 2021.[134]
Meta information on the timeline
How the timeline was built
The initial version of the timeline was written by Sebastian Sanchez.
Funding information for this timeline is available.
Feedback and comments
Feedback for the timeline can be provided at the following places:
- FIXME
What the timeline is still missing
- Updated visual data
- for books: https://academic-accelerator.com/encyclopedia/optimal-design
- doi: 10.1007/978-3-319-33781-4_1
- experiment design/design of experiments "in 1800..2020"
- Add Google Scholar table
- Vipul: "will this timeline eventually talk of things like double-blinding, triple-blinding, placebos, RCTs, etc., right? You have blinding but I guess the rest are variants on the idea".
- Vipul: "Cover "Statistical significance", "p-values" and preregistration."
- Books
- Placebo in history
- Glossary of experimental design
- Category:Design of experiments
- Design of experiments (check See also list)
- https://repository.rothamsted.ac.uk/id/eprint/29401/
- https://www.studocu.vn/vn/document/truong-dai-hoc-can-tho/phan-tich-va-thiet-ke-thuat-toan/15-a-brief-history-of-statistical-design-in-experiments-dae/139418259
- https://www.sciencedirect.com/topics/social-sciences/experimental-design
- [1]
- [2]
- [3]
- [4]
Timeline update strategy
See also
External links
References
- ↑ Yao, Mengxuan; Wang, Haicheng; Chen, Wei (May 2023). "Clinical research–When it matters". Injury. 54 (Supplement 3): S35 – S38. doi:10.1016/j.injury.2022.01.049. Retrieved April 9, 2025.
- ↑ Kleisiaris, Christos F.; Sfakianakis, Chrisanthos; Papathanasiou, Ioanna V. (March 15, 2014). "Health care practices in ancient Greece: The Hippocratic ideal". Journal of Medical Ethics and History of Medicine. 7. PMC 4263393. PMID 25512827. Retrieved April 9, 2025.
- ↑ "Baconian method". Encyclopaedia Britannica. Retrieved April 9, 2025.
- ↑ Lienhard, John H. (January 4, 1989). "Galileo's Experiment". The Engines of Our Ingenuity. University of Houston. Retrieved April 9, 2025.
- ↑ "Robert Boyle". Internet Encyclopedia of Philosophy. Retrieved April 9, 2025.
- ↑ Ore, Øystein (May 1960). "Pascal and the Invention of Probability Theory" (PDF). The American Mathematical Monthly. 67 (5). Mathematical Association of America: 409–419. JSTOR 2309286. Retrieved April 9, 2025.
- ↑ "Fermat and Pascal on Probability" (PDF). University of York. Retrieved April 9, 2025.
- ↑ Polasek, Wolfgang (August 2000). "The Bernoullis and the Origin of Probability Theory: Looking back after 300 Years". Resonance – Journal of Science Education. 5 (8): 26–42. Retrieved April 9, 2025.
- ↑ Cline, Douglas (August 9, 2020). "Age of Enlightenment". Physics LibreTexts. University of Rochester. Retrieved April 9, 2025.
- ↑ "The 'father of modern statistics' honoured". BBC News. September 9, 2016. Retrieved April 9, 2025.
- ↑ Schulz, Kathryn (August 14, 2023). "How Carl Linnaeus Set Out to Label All of Life". The New Yorker. Retrieved April 9, 2025.
- ↑ Schwarz, K. A., & Pfister, R.: Scientific psychology in the 18th century: a historical rediscovery. In: Perspectives on Psychological Science, Nr. 11, p. 399-407.
- ↑ "Statement on R A Fisher". Rothamsted Research. June 2020. Retrieved April 9, 2025.
- ↑ 14.00 14.01 14.02 14.03 14.04 14.05 14.06 14.07 14.08 14.09 14.10 14.11 14.12 "1.1 - A Quick History of the Design of Experiments (DOE) | STAT 503". PennState: Statistics Online Courses. Retrieved 11 May 2021.
- ↑ Preece, D. A. (December 1990). "R. A. Fisher and Experimental Design: A Review". Biometrics. 46 (4). International Biometric Society: 925–935. doi:10.2307/2532438. JSTOR 2532438. Retrieved April 9, 2025.
- ↑ Biau, David Jean; Jolles, Brigitte M.; Porcher, Raphaël (March 2010). "P Value and the Theory of Hypothesis Testing: An Explanation for New Researchers". Clinical Orthopaedics and Related Research. 468 (3): 885–892. doi:10.1007/s11999-009-1164-4. PMC 2816758. PMID 19921345. Retrieved April 9, 2025.
- ↑ Kramer, Lloyd; Maza, Sarah (23 June 2006). A Companion to Western Historical Thought. Wiley. ISBN 978-1-4051-4961-7.
Shortly after the start of the Cold War [...] double-blind reviews became the norm for conducting scientific medical research, as well as the means by which peers evaluated scholarship, both in science and in history.
- ↑ 18.0 18.1 "Clinical trials—from ancient Babylon to today". Main Line Health. 17 May 2021. Retrieved 31 March 2026.
- ↑ Aligabi, Zahra (2020). "Reflections on Avicenna's impact on medicine: his reach beyond the Middle East". Journal of Community Hospital Internal Medicine Perspectives. 10 (4): 310–312. doi:10.1080/20009666.2020.1774301. Retrieved 31 March 2026.
- ↑ Pearce, J. M. S. (29 September 2022). "Francis Bacon's natural philosophy and medicine". Hektoen International: A Journal of Medical Humanities. Retrieved 31 March 2026.
- ↑ Boyle, Robert (1683). New Experiments and Observations Touching Cold, or, An Experimental History of Cold, Begun. London: Richard Davis. Retrieved 31 March 2026.
- ↑ Colbourn, Charles J.; Dinitz, Jeffrey H. Handbook of Combinatorial Designs (2nd ed.). CRC Press. p. 12. ISBN 9781420010541. Retrieved 28 March 2017.
- ↑ "Choi Seok-jeong (1646–1715)". MacTutor History of Mathematics Archive. University of St Andrews. Retrieved 31 March 2026.
- ↑ 24.0 24.1 Dunn, Peter M. (January 1, 1997). "James Lind (1716-94) of Edinburgh and the treatment of scurvy". Archives of Disease in Childhood: Fetal and Neonatal Edition. 76 (1): F64–5. doi:10.1136/fn.76.1.F64. PMC 1720613. PMID 9059193.
- ↑ 25.0 25.1 25.2 25.3 25.4 25.5 25.6 "Designing of Experiments". StudyAndScore. Retrieved 9 April 2026.
- ↑ "Kent Academic Repository" (PDF). kar.kent.ac.uk. Retrieved 23 October 2021.
- ↑ "Carl Friedrich Gauss & Adrien-Marie Legendre Discover the Method of Least Squares". History of Information. Jeremy M. Norman. Retrieved 31 March 2026.
- ↑ "Polynomial regression". frontend. Retrieved 18 March 2022.
- ↑ Fétis, François-Joseph (1868). Biographie Universelle des Musiciens et Bibliographie Générale de la Musique, Tome 1 (Second ed.). Paris: Firmin Didot Frères, Fils, et Cie. p. 249. Retrieved 2011-07-21.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Dubourg, George (1852). The Violin: Some Account of That Leading Instrument and its Most Eminent Professors... (Fourth ed.). London: Robert Cocks and Co. pp. 356–357. Retrieved 2011-07-21.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Stigler (1986, pp 154–155)
- ↑ Stolberg, M. (December 2006). "Inventing the randomized double-blind trial: the Nuremberg salt test of 1835". Journal of the Royal Society of Medicine. 99 (12): 642–643. doi:10.1258/jrsm.99.12.642. PMC 1676327. PMID 17139070.
- ↑ Faerstein, Eduardo; Winkelstein Jr., Warren (September 2012). "Adolphe Quetelet: Statistician and More". Epidemiology. 23 (5): 762–763. doi:10.1097/EDE.0b013e318261c86f. Retrieved 31 March 2026.
- ↑ 34.0 34.1 "Experimental Psychology: History, Features, and Method". Psychologs Magazine. Retrieved April 9, 2025.
- ↑ Pasteur, Louis (1861). Sur les corpuscules organisés qui existent dans l'atmosphère: Examen de la doctrine des générations spontanées (in français). Paris: Ch. Lahure et Cie. Retrieved 31 March 2026.
- ↑ Cavaillon, Jean-Marc; Legout, Sandra (2022). "Louis Pasteur: Between Myth and Reality". Biomolecules. 12 (4): 596. doi:10.3390/biom12040596. PMC 9027159. PMID 35454184. Retrieved 31 March 2026.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ↑ Peirce, C. S. (August 1967). "Note on the Theory of the Economy of Research". Operations Research. 15 (4): 643–648. doi:10.1287/opre.15.4.643.
- ↑ Robert Burch (2001). "Charles Sanders Peirce". Stanford Encyclopedia of Philosophy. Metaphysics Research Lab, Stanford University. Retrieved 6 April 2026.
- ↑ Peirce, C. S. (1882), "Introductory Lecture on the Study of Logic" delivered September 1882, published in Johns Hopkins University Circulars, v. 2, n. 19, pp. 11–12, November 1882, see p. 11, Google Books Eprint. Reprinted in Collected Papers v. 7, paragraphs 59–76, see 59, 63, Writings of Charles S. Peirce v. 4, pp. 378–82, see 378, 379, and The Essential Peirce v. 1, pp. 210–14, see 210–1, also lower down on 211.
- ↑ Stigler (1986, pp 314–315)
- ↑ Charles Sanders Peirce and Joseph Jastrow (1885). "On Small Differences in Sensation". Memoirs of the National Academy of Sciences. 3: 73–83. http://psychclassics.yorku.ca/Peirce/small-diffs.htm
- ↑ Stroebe, W. (2012). The truth about Triplett (1898), but nobody seems to care. Perspectives on Psychological Science, 7, 54-57.
- ↑ Pearson, Karl (1900). "On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling" (PDF). Philosophical Magazine. Series 5. 50 (302): 157–175. doi:10.1080/14786440009463897.
- ↑ Nahm, Francis Sahngun (2017). "What the P values really tell us". The Korean Journal of Pain. 30 (4): 241. doi:10.3344/kjp.2017.30.4.241.
- ↑ Nahm, Francis Sahngun (October 2017). "What the P values really tell us". The Korean Journal of Pain. 30 (4): 241–242. doi:10.3344/kjp.2017.30.4.241. ISSN 2005-9159.
- ↑ Newman, David H., M.D. (2008). Hippocrates' shadow : secrets from the house of medicine (1st Scribner hardcover ed.). New York, NY: Scribner. ISBN 978-1-4165-5153-9.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ Rivers WH, Webber HN (August 1907). "The action of caffeine on the capacity for muscular work". The Journal of Physiology. 36 (1): 33–47. doi:10.1113/jphysiol.1907.sp001215. PMC 1533733. PMID 16992882.
- ↑ Gimhan, Vishal (6 March 2025). "Understanding the t-Distribution: A Guide for Small Sample Analysis". Medium. Retrieved 31 March 2026.
- ↑ 49.0 49.1 49.2 49.3 49.4 49.5 49.6 49.7 49.8 "Chapter 2 A Brief History of Experimental Design". JABSTB: Statistical Design and Analysis of Experiments with R. Retrieved 2026-04-07.
- ↑ The Correlation Between Relatives on the Supposition of Mendelian Inheritance. Ronald A. Fisher. Philosophical Transactions of the Royal Society of Edinburgh. 1918. (volume 52, pages 399–433)
- ↑ Smith, Kirstine (1918). "On the Standard Deviations of Adjusted and Interpolated Values of an Observed Polynomial Function and its Constants and the Guidance they give Towards a Proper Choice of the Distribution of Observations". Biometrika. 12 (1–2). Oxford University Press: 1–85. doi:10.2307/2331929. Retrieved 31 March 2026.
- ↑ 52.0 52.1 "Experimental Design | Encyclopedia.com". www.encyclopedia.com. Retrieved 5 April 2021.
- ↑ On the "Probable Error" of a Coefficient of Correlation Deduced from a Small Sample. Ronald A. Fisher. Metron, 1: 3–32 (1921)
- ↑ 54.00 54.01 54.02 54.03 54.04 54.05 54.06 54.07 54.08 54.09 54.10 54.11 54.12 54.13 54.14 54.15 54.16 54.17 Chandramouli, R. "100 years of DoE – from Fisher to Jones". LinkedIn. Retrieved 2026-04-07.
- ↑ 55.00 55.01 55.02 55.03 55.04 55.05 55.06 55.07 55.08 55.09 55.10 "1.5 A Brief History of Statistical Design in Experiments (DAE)". Studocu. Retrieved 2026-04-07.
- ↑ Scheffé (1959, p 291, "Randomization models were first formulated by Neyman (1923) for the completely randomized design, by Neyman (1935) for randomized blocks, by Welch (1937) and Pitman (1937) for the Latin square under a certain null hypothesis, and by Kempthorne (1952, 1955) and Wilk (1955) for many other designs.")
- ↑ Cumming, Geoff (2011). "From null hypothesis significance to testing effect sizes". Understanding The New Statistics: Effect Sizes, Confidence Intervals, and Meta-Analysis. Multivariate Applications Series. East Sussex, United Kingdom: Routledge. pp. 21–52. ISBN 978-0-415-87968-2.
- ↑ Fisher, Ronald A. (1925). Statistical Methods for Research Workers. Edinburgh, UK: Oliver and Boyd. pp. 43. ISBN 978-0-050-02170-5.
{{cite book}}: ISBN / Date incompatibility (help) - ↑ Lehmann, Erich L. (2011). Fisher, Neyman, and the creation of classical statistics. New York, NY: Springer Science+Business Media, LLC. p. 15. ISBN 978-1-4419-9500-1.
- ↑ Conniffe, Denis (1990–1991). "R. A. Fisher and the development of statistics—a view in his centenary year". Journal of the Statistical and Social Inquiry Society of Ireland. Vol. XXVI, no. 3. Dublin: Statistical and Social Inquiry Society of Ireland. p. 87. hdl:2262/2764. ISSN 0081-4776.
- ↑ Savage, Leonard J. (1976). "On Rereading R. A. Fisher". Annals of Statistics. 4 (3): 441–500. doi:10.1214/aos/1176343456.
- ↑ Kopf, Dan. "An error made in 1925 led to a crisis in modern science—now researchers are joining to fix it". Quartz. Retrieved 13 March 2021.
- ↑ 63.00 63.01 63.02 63.03 63.04 63.05 63.06 63.07 63.08 63.09 63.10 63.11 63.12 63.13 63.14 63.15 63.16 "Experimental Design". Encyclopedia.com. Gale. Retrieved 2026-04-07.
- ↑ Fisher, Ronald. "The Arrangement of Field Experiments" (PDF). Journal of the Ministry of Agriculture of Great Britain. London, England: Ministry of Agriculture and Fisheries.
- ↑ Box, Joan Fisher (February 1980). "R. A. Fisher and the Design of Experiments, 1922-1926". The American Statistician. 34 (1): 1. doi:10.2307/2682986.
- ↑ Box, JF (February 1980). "R. A. Fisher and the Design of Experiments, 1922–1926". The American Statistician. 34 (1): 1–7. doi:10.2307/2682986. JSTOR 2682986.
- ↑ Yates, F (June 1964). "Sir Ronald Fisher and the Design of Experiments". Biometrics. 20 (2): 307–321. doi:10.2307/2528399. JSTOR 2528399.
- ↑ Stanley, Julian C. (1966). "The Influence of Fisher's "The Design of Experiments" on Educational Research Thirty Years Later". American Educational Research Journal. 3 (3): 223–229. doi:10.3102/00028312003003223. JSTOR 1161806.
- ↑ "Completely Randomized Design". TheFreeDictionary.com. Retrieved 16 March 2021.
- ↑ Bose, R. C.; Nair, K. R. (1939), "Partially balanced incomplete block designs", Sankhyā, 4: 337–372
- ↑ "5.3.3.5. Plackett-Burman designs". www.itl.nist.gov. Retrieved 22 July 2023.
- ↑ "The MRC randomized trial of streptomycin and its legacy". PubMed Central. Retrieved 3 April 2026.
- ↑ Healy, M. J. R. (1995). "Frank Yates, 1902-1994: The Work of a Statistician". International Statistical Review / Revue Internationale de Statistique. 63 (3): 271–288. ISSN 0306-7734.
- ↑ Grundy, P. M.; Healy, M. J. R. (1950). "Restricted Randomization and Quasi-Latin Squares". Journal of the Royal Statistical Society. Series B (Methodological). 12 (2): 286–291. ISSN 0035-9246.
- ↑ Template:Cite thesis
- ↑ "Experimental Designs". www.amazon.com.
- ↑ Draper, Norman R. (1992). "Introduction to Box and Wilson (1951) On the Experimental Attainment of Optimum Conditions". Breakthroughs in Statistics: Methodology and Distribution. Springer. pp. 267–269. doi:10.1007/978-1-4612-4380-9_22.
- ↑ Robbins, Herbert (1952). "Some aspects of the sequential design of experiments". Bulletin of the American Mathematical Society. 58 (5): 527–535. doi:10.1090/S0002-9904-1952-09620-8.
- ↑ Bose, R. C.; Shimamoto, T. (June 1952). "Classification and Analysis of Partially Balanced Incomplete Block Designs with Two Associate Classes". Journal of the American Statistical Association. 47 (258): 151–184. doi:10.1080/01621459.1952.10501161.
- ↑ Hróbjartsson A, Gøtzsche PC (May 2001). "Is the placebo powerless? An analysis of clinical trials comparing placebo with no treatment". The New England Journal of Medicine. 344 (21): 1594–602. doi:10.1056/NEJM200105243442106. PMID 11372012.
- ↑ "Genichi Taguchi". asq.org. American Society for Quality. Retrieved 31 March 2026.
- ↑ Ranade, Shruti Sunil; Thiagarajan, Padma (November 2017). "Selection of a design for response surface". IOP Conference Series: Materials Science and Engineering. 263: 022043. doi:10.1088/1757-899X/263/2/022043.
- ↑ Kish, Leslie (1965). "Survey Sampling". New York: John Wiley & Sons, Inc. ISBN 0-471-10949-5.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "Definition of NOCEBO". www.merriam-webster.com. Retrieved 5 March 2022.
- ↑ Kennedy, 1961
- ↑ Smith, Vernon L. (1962). "An Experimental Study of Competitive Market Behavior". Chapman University Digital Commons. Chapman University. Retrieved 31 March 2026.
- ↑ Stankova, Tatiana (30 June 2020). "Application of Nelder wheel experimental design in forestry research". Silva Balcanica. 21 (1): 29–40. doi:10.3897/silvabalcanica.21.e54425.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ↑ 88.0 88.1 Heppner, Puncky Paul; Wampold, Bruce E.; Owen, Jesse; Wang, Kenneth T. (21 August 2015). Research Design in Counseling. Cengage Learning. ISBN 978-1-305-46501-5.
- ↑ "Textbooks and other publications on controlled clinical trials". PubMed Central. Retrieved 3 April 2026.
- ↑ "Design of experiments". Wikipedia. Retrieved 3 April 2026.
- ↑ Chernoff, H. (1972) Sequential Analysis and Optimal Design, SIAM Monograph.
- ↑ Montgomery, Douglas C. (2013). Design and Analysis of Experiments. John Wiley & Sons Incorporated. ISBN 978-1-62198-227-2.
- ↑ Pocock S (2005). "When (not) to stop a clinical trial for benefit" (PDF). JAMA. 294 (17): 2228–2230. doi:10.1001/jama.294.17.2228. PMID 16264167.
- ↑ "Experimental Design - an overview | ScienceDirect Topics". www.sciencedirect.com. Retrieved 23 March 2021.
- ↑ Ma, Will. "Lecture 1: Four (and a Half) Proofs of the Basic Prophet Inequality" (PDF). Columbia University. Columbia Business School. Retrieved 31 March 2026.
- ↑ Richter, Felicitas; Dewey, Marc (September 2014). "Zelen Design in Randomized Controlled Clinical Trials". Radiology. 272 (3): 919–919. doi:10.1148/radiol.14140834.
- ↑ Homer, Caroline S.E. (April 2002). "Using the Zelen design in randomized controlled trials: debates and controversies". Journal of Advanced Nursing. 38 (2): 200–207. doi:10.1046/j.1365-2648.2002.02164.x.
- ↑ McKay, M.D.; Beckman, R.J.; Conover, W.J. (May 1979). "A Comparison of Three Methods for Selecting Values of Input Variables in the Analysis of Output from a Computer Code". Technometrics. 21 (2). American Statistical Association: 239–245. doi:10.2307/1268522. ISSN 0040-1706. JSTOR 1268522.
- ↑ "Design of experiments". Wikipedia. Retrieved 3 April 2026.
- ↑ "Randomized controlled trial - History". Wikipedia. Retrieved 3 April 2026.
- ↑ Karkar, Ravi; Zia, Jasmine; Vilardaga, Roger; Mishra, Sonali R; Fogarty, James; Munson, Sean A; Kientz, Julie A (1 May 2016). "A framework for self-experimentation in personalized health". Journal of the American Medical Informatics Association. 23 (3): 440–448. doi:10.1093/jamia/ocv150.
- ↑ George E.P., Box (2006). Improving Almost Anything: Ideas and Essays (Revised ed.). Hoboken, New Jersey: Wiley.
- ↑ "Revisiting Hurlbert 1984". Reflections on Papers Past. 29 November 2020. Retrieved 29 March 2022.
- ↑ LaLonde, Robert (1986). "Evaluating the Econometric Evaluations of Training Programs with Experimental Data". American Economic Review. 4 (76): 604–620.
- ↑ Street, Anne Penfold; Street, Professor of Mathematics Anne Penfold; Street, Deborah J.; Street, Lecturer in Biometry Deborah J. (1987). "Combinatorics of Experimental Design". Clarendon Press.
- ↑ Mead, R. (26 July 1990). The Design of Experiments: Statistical Principles for Practical Applications. Cambridge University Press. ISBN 978-0-521-28762-3.
- ↑ Haaland, Perry D. (1989). Experimental design in biotechnology. New York: Marcel Dekker. ISBN 9780824778811.
- ↑ Haaland, Perry D. (June 1991). "BOOK REVIEW: EXPERIMENTAL DESIGN IN BIOTECHNOLOGY Perry D. Haaland Marcel Dekkwe, Inc., New York, 1989". Drying Technology. 9 (3): 817–817. doi:10.1080/07373939108916715.
- ↑ Haaland, Perry D. (25 November 2020). "Experimental Design in Biotechnology". doi:10.1201/9781003065968.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "Read "Statistical Methods for Testing and Evaluating Defense Systems: Interim Report" at NAP.edu". Retrieved 14 March 2021.
- ↑ "Evidence-based medicine". Wikipedia. Retrieved 3 April 2026.
- ↑ Horne, G., & Schwierz, K. (2008). Data farming around the world overview. Paper presented at the 1442-1447. doi:10.1109/WSC.2008.4736222
- ↑ Neyer, Barry T. (February 1994). "A D-Optimality-Based Sensitivity Test". Technometrics. 36 (1): 61. doi:10.2307/1269199.
- ↑ "Integrated Addendum to ICH E6(R1): Guideline for Good Clinical Practice E6(R2)" (PDF). International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. 9 November 2016. Retrieved 31 March 2026.
- ↑ "Randomized controlled trial - CONSORT". Wikipedia. Retrieved 3 April 2026.
- ↑ Li He, "Design of Experiments Software, DOE software", The Chemical Information Network, July 17, 2003.
- ↑ "Analyzing Families of Experiments in SE: a Systematic Mapping Study" (PDF). arxiv.org. Retrieved 12 March 2022.
- ↑ Oehlert, Gary W. (2010). A First Course in Design and Analysis of Experiments. Gary W. Oehlert.
- ↑ "Randomized controlled trial - applications". Wikipedia. Retrieved 3 April 2026.
- ↑ "Adversarial Collaboration: An EDGE Lecture by Daniel Kahneman | Edge.org". www.edge.org. Retrieved 8 March 2022.
- ↑ Schneider, ed. by Sandra L.; Shanteau, James (2003). Emerging perspectives on judgment and decision research. Cambridge [u.a.]: Cambridge Univ. Press. pp. 438–9. ISBN 052152718X.
{{cite book}}:|first=has generic name (help) - ↑ "Randomized controlled trials in development economics". Wikipedia. Retrieved 3 April 2026.
- ↑ Pildal J, Chan AW, Hróbjartsson A, Forfang E, Altman DG, Gøtzsche PC (2005). "Comparison of descriptions of allocation concealment in trial protocols and the published reports: cohort study". BMJ. 330 (7499): 1049. doi:10.1136/bmj.38414.422650.8F. PMC 557221. PMID 15817527.
- ↑ Kahneman, Daniel; Klein, Gary. Conditions for intuitive expertise: A failure to disagree. American Psychologist, Vol 64(6), Sep 2009, 515-526. doi: 10.1037/a0016755
- ↑ Wagenmakers, E.-J., Wetzels, R., Borsboom, D., & van der Maas, H. L. J. (2010). Why psychologists must change the way they analyze their data: The case of psi.
- ↑ Hróbjartsson A, Gøtzsche PC (January 2010). Hróbjartsson A (ed.). "Placebo interventions for all clinical conditions" (PDF). The Cochrane Database of Systematic Reviews. 106 (1): CD003974. doi:10.1002/14651858.CD003974.pub3. PMID 20091554.
- ↑ "Randomized controlled trial - applications". Wikipedia. Retrieved 3 April 2026.
- ↑ Nosek, Brian A.; Ebersole, Charles R.; DeHaven, Alexander C.; Mellor, David T. (13 March 2018). "The preregistration revolution". Proceedings of the National Academy of Sciences. 115 (11): 2600–2606. doi:10.1073/pnas.1708274114.
- ↑ "Nobel Prize 2019 experimental approach to poverty". Wikipedia. Retrieved 3 April 2026.
- ↑ "Adaptive designs for clinical trials of drugs and biologics: Guidance for industry". U.S. Food and Drug Administration (FDA). 1 November 2019. Retrieved 7 April 2021.
- ↑ "Image-based treatment effect heterogeneity". arXiv. Retrieved 3 April 2026.
- ↑ "Design of experiments". Google Trends. Retrieved 14 December 2021.
- ↑ "Design of experiments". books.google.com. Retrieved 14 December 2021.
- ↑ "Design of experiments". wikipediaviews.org. Retrieved 14 December 2021.